16 Oct, 2025
The pharmaceutical industry’s commitment to producing safe, sterile medications relies heavily on advanced quality control tools, among which
biological indicators (BIs) stand out as a cornerstone of sterilization validation. As regulatory standards tighten and patient safety remains paramount,
BIs have become indispensable in verifying the effectiveness of sterilization processes during drug production.
What Are Biological Indicators?
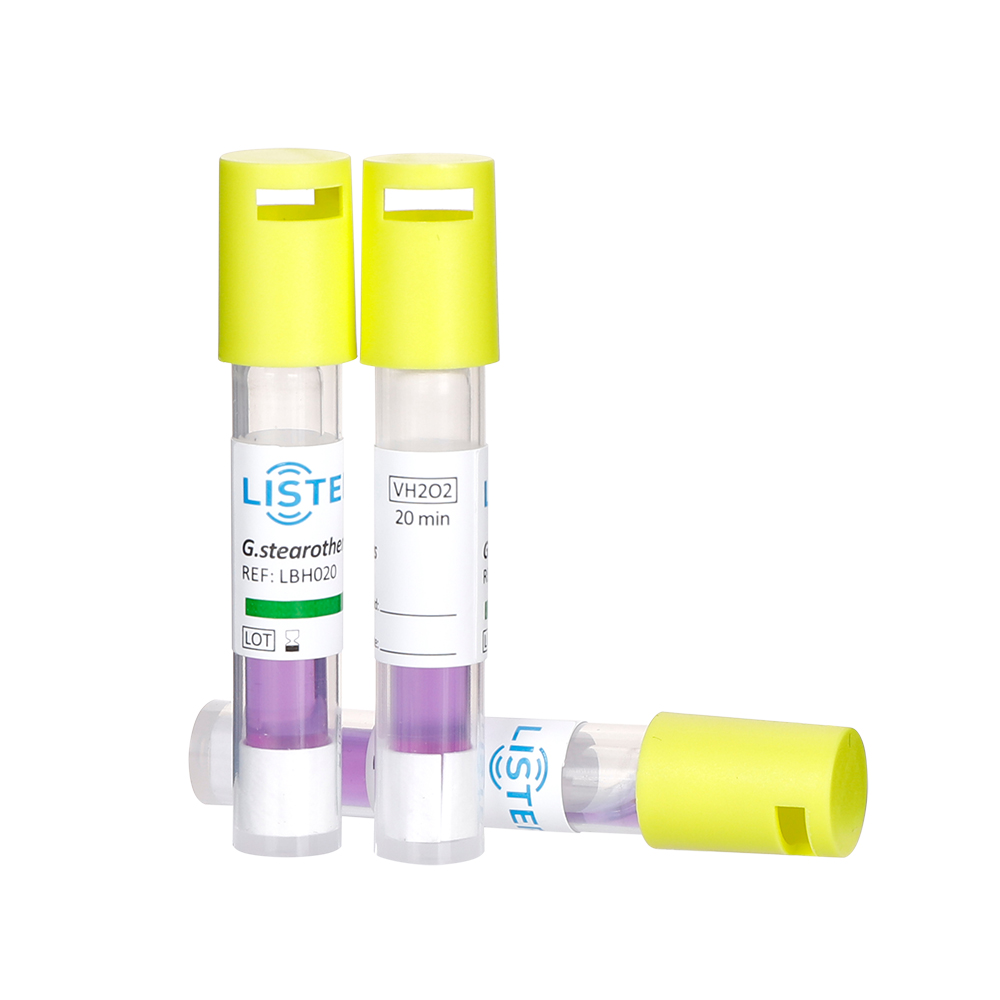
Biological indicators are standardized preparations of highly resistant microorganisms, such as bacterial spores (Geobacillus stearothermophilus
for steam sterilization or Bacillus atrophaeus for ethylene oxide), used to challenge sterilization systems. Their ability to survive under extreme
conditions provides direct evidence of whether a sterilization cycle achieves the required microbial kill rate.
Key Benefits of BIs in Pharma Manufacturing
Reliable Sterilization Assurance:
Unlike chemical indicators, which only confirm physical parameters (e.g., temperature or pressure), BIs offer definitive proof that a sterilization
process eliminates even the most resistant microbes. This is critical for injectables, implants, and other sterile products.
Regulatory Compliance:
Global regulatory bodies, including the FDA and EMA, mandate the use of BIs to validate sterilization methods. Their use ensures compliance
with pharmacopeial standards (e.g., USP <1229> and ISO 11138), reducing the risk of product recalls or regulatory penalties.
Risk Mitigation:
Contaminated pharmaceutical products pose life-threatening risks. BIs act as a fail-safe mechanism, preventing under-sterilized batches from
reaching patients. For example, in autoclave validation, a "BI failure" immediately flags process deviations, enabling corrective actions.
Process Optimization:
Data from BI testing helps manufacturers refine sterilization protocols, minimizing cycle times without compromising efficacy. This enhances
production efficiency and reduces energy costs.
Versatility:
BIs are tailored to diverse sterilization methods—steam, dry heat, radiation, or gas—making them adaptable to various drug manufacturing workflows.
Industry Trends and Innovations
Recent advancements include rapid-readout BIs, which provide results in hours instead of days, and self-contained indicators with integrated
growth media. Such innovations accelerate release timelines for critical products like vaccines and biologics. Additionally, digital tracking systems
now log BI data electronically, improving traceability for audits.
Conclusion
As pharmaceutical manufacturing evolves, biological indicators remain a non-negotiable tool for ensuring product safety and regulatory adherence.
LISTER BIOMEDICAL CO.,LTD By combining cutting-edge technology with rigorous microbial testing, the industry continues to uphold its promise
of delivering sterile, life-saving therapies to patients worldwide.
Keywords: Biological indicators, sterilization validation, pharmaceutical manufacturing.
Tags: